To Prepare Aluminium Hydroxide , [Al(OH)3] sol
Theory
Aluminium hydroxide sol is hydrophobic in nature. It is obtained by hydrolysis of aluminium chloride
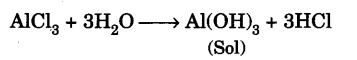
Dialysis is done to remove hydrochloric acid (produced as a result of hydrolysis of aluminium chloride) because aluminium hydroxide sol is affected by the presence of ionic
impurities.
Apparatus
Conical flask (250 ml), beaker (250 ml), a boiling-tube, glass-rod, funnel, round-bottom flask, iron stand with a clamp, wire-gauze, tripod-stand, burner and a burette or a dropper.
Materials Required
Aluminium chloride (2% solution) and distilled water.
Procedure
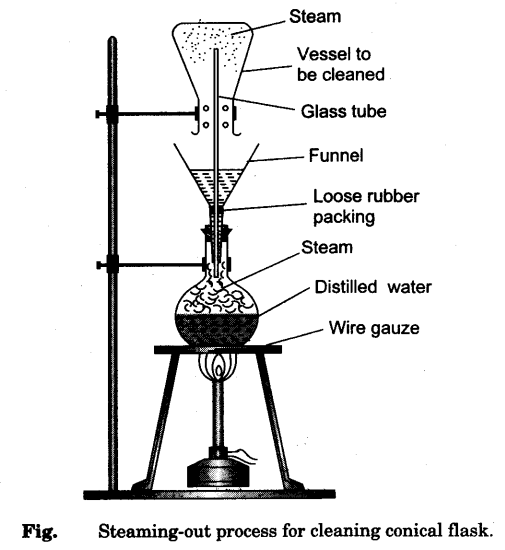
- Take a 250 ml conical flask and clean it by steaming-out process as shown in Fig.
- To this cleaned flask, add 100 ml of distilled water and heat it to boil by placing the flask on a wire-gauze.
- Add ferric chloride solution dropwise (by the use of a burette or a dropper) to the boiling water.
- Continue heating until deep red or brown solution of ferric hydroxide is obtained. Replace the water lost by evaporation during boiling at regular intervals.
- Keep the contents of conical flask undisturbed for sometime at room temperature. Label the solution as “ferric hydroxide sol”.
Chemistry Lab ManualNCERT Solutions Class 12 Chemistry Sample Papers
<!–
–>